Breath-Based Diagnostics
Diagnostics as easy as breathing
Interested in our exhaled breath condensate (EBC) collection kit?

Simple, Accessible, Affordable Mask-Based Testing
DiagMetrics is the first and only company in the world to develop a Mask-Based Diagnostic (MBDTM) collecting exhaled breath condensate (EBC) as a simple and non-invasive sample for rapid diagnostic testing.
Our EBC testing technology is versatile and adaptable to various testing platforms ranging the low cost and rapid lateral flow assay (LFA) to highly sensitive multiplexed semiconductor smart biosensor for remote patient monitoring.
Our first product will be a rapid TB screening LFA, designed with our partner MilliporeSigma, for remote point-of-care tuberculosis screening using EBCs collected by a patient in under 5 minutes.
DiagMetrics teaming up with MilliporeSigma to develop low-cost, rapid lateral flow assay (LFA) screening for active tuberculosis.
What is EBC?
Exhaled breath condensate (EBC) is the liquid formed when the water vapor and tiny aerosolized droplets from a person’s exhaled breath are cooled and collected. It contains various biomarkers—such as proteins, and inflammatory mediators—that originate from the respiratory tract and reflect lung and systemic health. EBC collection is noninvasive and is often used in research to study respiratory diseases, environmental exposures, and metabolic processes.
Through our patented EBC collection mask, DiagMetrics is making diagnostic as easy as 1-2-3 through Mask Based Diagnostic (MBD™).
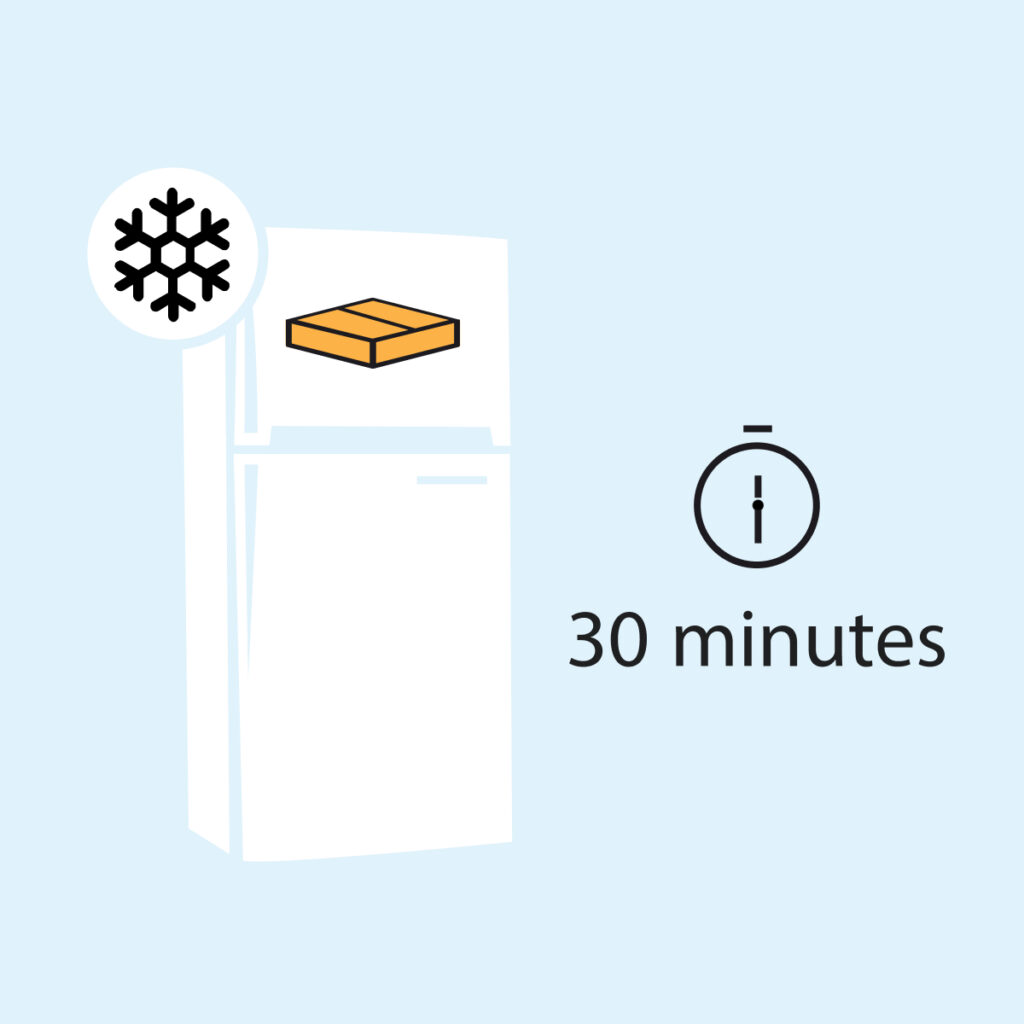
1. Freeze

2. Breathe

3. Test
Revolutionize Diagnostics with MBDTM Our 3-Prong Market Entry
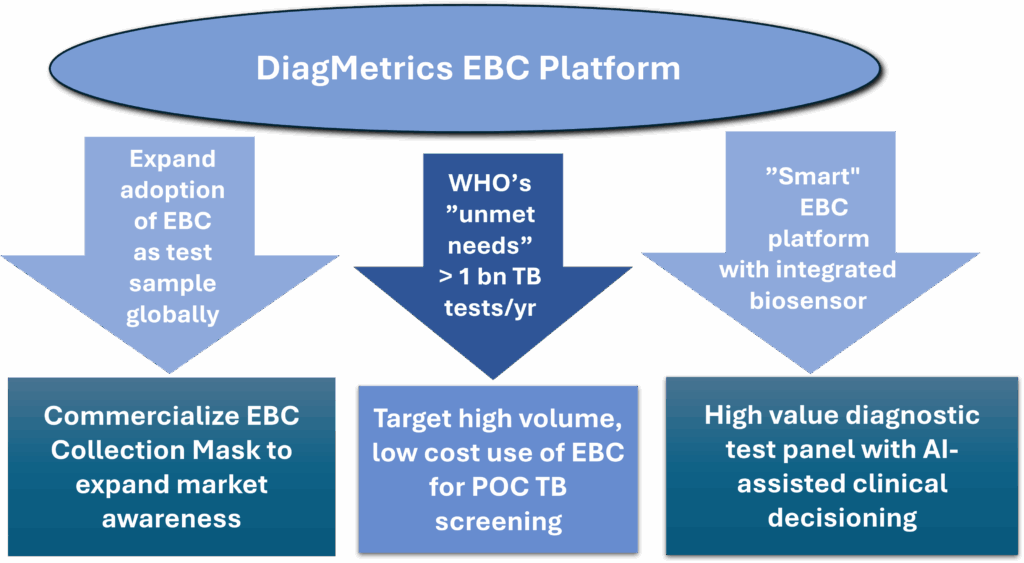
- Research Use Only (RUO) EBC collection mask deployment will accelerate adoption of breath based rapid testing worldwide
- Our technology is ready for simultaneous multiple biomarker panel tests such as SARS/Flu A/B/RSV
- Targeting identification of active & latent cases of tuberculosis which infects 23% of the global population
- Biomarkers in EBC can potentially be used for a variety of conditions such as:
- Early detection of respiratory cancers
- Monitoring chronic illness for changes warranting immediate treatment
- Confirming the extent of medication compliance
Our Intellectual Property
DiagMetrics’ aggressive IP protection strategy aims at building a world-class U.S. and International Patent Portfolio.
- Five U.S. patents issued, 14 U.S. patents pending, 4 PCT applications
- Extensive patent disclosures cover diagnostic device construction, methods of manufacturing, electronic and non-electronic biosensors, disease-specific use-cases, remote patient monitoring, AI-assisted diagnosis, and alternatives for EBC collection
- Five U.S. patents issued, 14 U.S. patents pending, 4 PCT applications
- Japan, India, China and South Korea, EPO, Canada, US, Mexico and Brazil
DiagMetrics News
Visit the news center for the most recent product updates, publications, and announcements.

DiagMetrics Secures Key U.S. Patents Covering Breath-Based Diagnostics and Strengthens Intellectual Property Leadership
DiagMetrics, Inc. has been granted three U.S. patents — 12,369,816; 12,092,639; and 12,031,982 — covering its Mask-Based Diagnostic™ (MBD™) platform for collecting and analyzing exhaled breath condensate (EBC). These patents secure broad protection for the company’s innovations in EBC collection, diagnostic integration with rapid assays and biosensors, and thermal mass condensation technologies. The new IP strengthens DiagMetrics’ leadership in non-invasive, breath-based diagnostics, supporting its mission to deliver affordable, scalable solutions for global health challenges including infectious disease, oncology, cardiology, and chronic disease monitoring

DiagMetrics Developing Capture Molecule- and Biosensor-Based Breath Assays
Startup DiagMetrics plans to parlay novel sample collection and detection technologies into home-use and point of care tests, and was recently awarded funding.
DiagMetrics Recieves NIH RADx® Funding
DiagMetrics has recieved an NIH RADx® award to advance its mask-based diagnostic platform.